“A life-changing opportunity” - More boys accessing clinical trials through the DMD Hub

The first boy in the UK to take part in Sarepta’s DMD gene therapy trial enjoyed a starring role in a presentation at the Action Duchenne conference last week.
Newcastle University’s Emma Heslop addressed the conference on the Duchenne UK funded DMD Hub, which aims to expand opportunities for UK patients to take part in clinical research.
Previously, clinical trials and studies were often turned away from the UK due to lack of capacity. Since the DMD Hub’s launch, hundreds of boys are now taking part in research across the UK.
She told the conference how Charlie’s mum Jennifer had filled his details onto the The DMD Hub Central Recruitment Pilot Project.
The project aims to create a national contact list of children and adults with DMD who are interested in participating in research studies. Sites running studies can access a wider pool of patients outside of their local area, meaning patients that don’t live near a site have the opportunity to take part, if eligible.
Earlier this summer Jennifer was taking her three kids to the dentist when the phone rang. It was Newcastle University, telling her that Charlie may be eligible to take part in a gene therapy trial.
Jennifer urged members of the DMD community to sign up:
“I’m very glad that the moment I saw the Facebook post about the DMD Hub’s central recruitment project, I thought – I’m going to do that now. I’ve constantly got this great long list of things to do, but without it they would not have had our details, and you would be speaking to some other little boy’s mum or dad – and not me. We would not have been given this potentially life-changing opportunity. We are very, very grateful.”
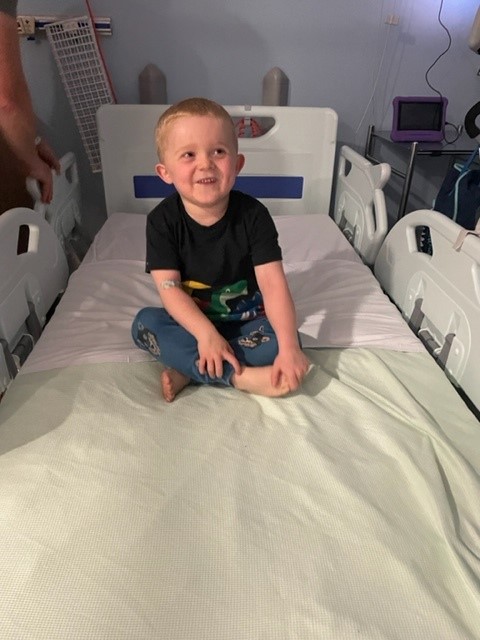
Charlie is taking part in the EMBARK study, a Phase 3 trial looking at the efficacy and safety of Sarepta’s gene transfer therapy in boys with DMD aged 4.
Find out more about taking part in clinical research on the DMD Hub’s website.